FDA: 'Influencers' promoted vaping without suitable warnings
WASHINGTON – U.S. regulators moved to discipline vaping companies for inappropriately promoting their flavored nicotine formulas through so-called influencers on Facebook, Twitter and other social media sites.
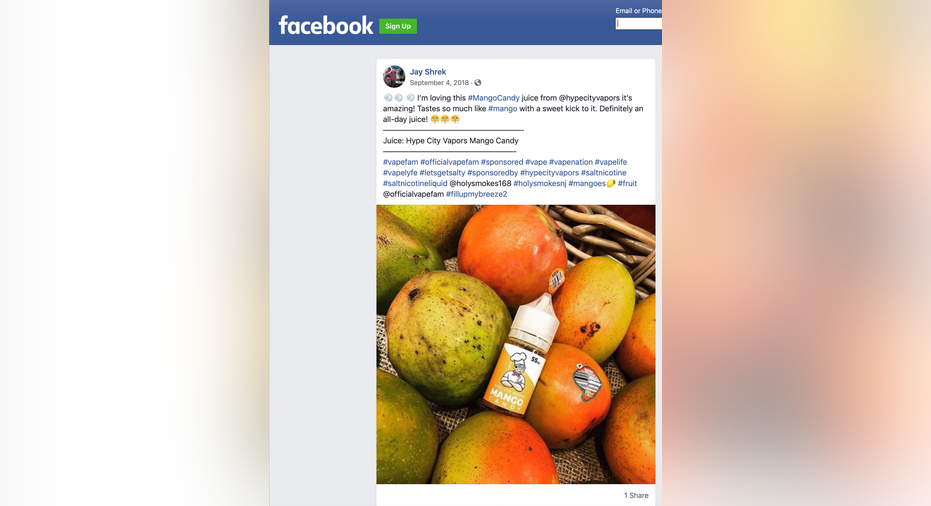
The Food and Drug Administration sent warning letters Friday to four companies that used paid social media influencers to pitch nicotine solutions to their online followers, including flavors like Watermelon Patch and Strawberry Kiwi.
The posts didn't include a mandatory warning that the vaping liquids contain nicotine, which is addictive. The FDA, joined by the Federal Trade Commission, sent the letters to Solace Vapor, Hype City Vapors, Humble Juice Co. and Artist Liquid Labs. The companies did not immediately return calls and emails seeking comment Friday morning.
Facebook prohibits e-cigarette ads even with warnings and the FTC has been pressuring influencers — people with many social media followers who promote products and services — to disclose when they are being paid to endorse something.
The action comes as the FDA and other government agencies struggle to reverse what they call an epidemic of underage e-cigarette use. Researchers have linked the trend to a surge in online videos, photos and other posts about vaping, some of them generated by companies, advertising agencies and paid influencers.
Government figures showed a nearly 80 percent jump in vaping by teens last year, with 1 in 5 high school students reporting that they used the devices in the previous month.
E-cigarettes typically heat a flavored nicotine solution into an inhalable aerosol. They are largely viewed as less harmful than traditional paper-and-tobacco cigarettes and some adult smokers use them as an alternative source of nicotine. But health experts warn that nicotine can harm developing brains and recent research shows many teenagers are unaware they are consuming the addictive chemical when they vape.
"It's critical we ensure manufacturers, retailers and others are including the required health warning about nicotine's addictive properties on packages and advertisements — especially on social media platforms popular with kids," acting FDA Commissioner Ned Sharpless said in a statement.
The FDA letters do not mention Juul, which dominates the U.S. e-cigarette market. The Silicon Valley startup is widely credited with helping trigger an explosion in vaping with its early viral marketing, which included paid posts and referrals by social media influencers.
After coming under intense scrutiny last year, Juul shuttered its Facebook and Instagram accounts in November. Since then, the company has reworked its marketing to focus on older adult smokers who are interested in quitting cigarettes.
In response to written questions from Senate lawmakers, Juul said in April it does not currently use paid social media influencers.
___
Follow Matthew Perrone on Twitter: @AP_FDAwriter
___
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute's Department of Science Education. The AP is solely responsible for all content.



















