Eye drops sold at CVS, Rite Aid, Target could cause eye infections and vision loss, FDA says
CVS, Rite Aid, Target already removing eye drop products after FDA warning
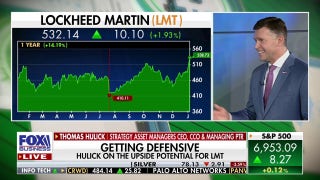
The Food and Drug Administration (FDA) is telling consumers to avoid two dozen eye drop products sold at major retailers including CVS and Rite Aid, because they could cause eye infections and potentially lead to vision loss.
The 26 products treat dry or irritated eyes and were marketed under the brands CVS Health, Leader (Cardinal Health), Rugby (Cardinal Health), Rite Aid, Velocity Pharma and Target's Up & Up brand. They pose a "potential risk of eye infections that could result in partial vision loss or blindness," according to the FDA.
The warnings from the FDA were prompted after investigators discovered that there were "insanitary conditions" in the manufacturer's facility. There were also positive bacterial test results from environmental sampling of critical drug production areas in the facility, the FDA said.
The FDA did not name the manufacturer, but CVS and Cardinal Health noted that the products in question were supplied by Velocity Pharma.
EYE DROP RECALL: WHAT IS PSEUDOMONAS AERUGINOSA BACTERIA?
Federal health officials said CVS, Rite Aid and Target are already removing the products from store shelves and their online marketplaces. However, regulators cautioned that "products branded as Leader, Rugby and Velocity may still be available to purchase in stores and online."
| Ticker | Security | Last | Change | Change % |
|---|---|---|---|---|
| CVS | CVS HEALTH CORP. | 75.77 | -2.58 | -3.29% |
| TGT | TARGET CORP. | 115.52 | -0.03 | -0.03% |
A spokesperson for CVS told FOX Business in a statement that it immediately stopped the sale of all products supplied by Velocity Pharma within the CVS Health Brand Eye Products portfolio after being notified by the FDA. CVS said that consumers who already purchased it could return the product for a refund.

A person uses a dropper to put artificial tears in their eye. (iStock / iStock)
"We’re committed to ensuring the products we offer are safe, work as intended and satisfy customers, and are fully cooperating with the FDA on this matter," the CVS spokesperson said.
Cardinal Health also told FOX Business that it "placed all identified impacted eye drop products in our inventory on hold" and is working with Velocity Pharma and FDA to initiate a recall of all impacted Rugby Laboratories and Cardinal Health Leader branded eye drop products.
"We are working with Velocity Pharma, the supplier of the impacted eye drop products to gain additional insight regarding the unsanitary conditions identified by the FDA at the manufacturing facility," Cardinal Health said in a statement.
Target, Rite Aid and Velocity Pharma did not immediately respond to FOX Business' request for comment.
EYE DROP RECALL: FDA FINDS STERILIZATION ISSUES AT GLOBAL HEALTH PHARMA FACILITY
The FDA told the manufacturers of the affected products to recall all lots on Oct. 25. The FDA did not specify the type of bacteria that was found during testing.
To date, the agency also has not tied this warning to the prior outbreak of antibiotic-resistant bacteria pseudomonas aeruginosa linked to eye products from Global Pharma Healthcare.
A close-up view of a person's eye. (Maciej Luczniewski/NurPhoto via Getty Images / Getty Images)
The products "are intended to be sterile." However, the FDA said that there is a "heightened risk of harm" with ophthalmic drug products, or products pertaining to the eye, due to the fact that drugs that are applied to the eyes bypass some of the body’s natural defenses.
There have not been any reports of eye infections related to these products to date. However, consumers who have signs or symptoms of an eye infection after using these products are told to seek medical care "immediately."
GET FOX BUSINESS ON THE GO BY CLICKING HERE
Symptoms of an eye infection include irritated or red eyes, worsening pain in or around the eyes – even after contact lens removal, light sensitivity, sudden blurry vision or unusually watery eyes or discharge, according to the Centers for Disease Control and Prevention.
Patients are encouraged to report any adverse events or quality problems they have with any medicine to the agency's MedWatch Adverse Event Reporting program.