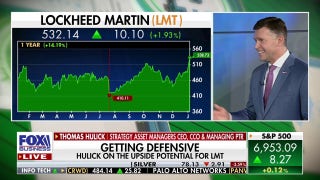
Whole Foods' private label nutritional supplement recalled over undeclared allergens
The containers were mistakenly packaged with Soy Protein Powder
A nutritional supplement packaged under Whole Foods' private label, 365 Everyday Value, is being recalled due to mislabeling and undeclared soy.
The manufacturer, Arizona Nutritional Supplements, voluntarily recalled 13.9-oz. containers of the 365 Everyday Value Whey Protein Powder Natural Vanilla Flavor "due to the potential of an undeclared soy allergen," the U.S. Food and Drug Administration (FDA) announced.
CLICK HERE TO READ MORE ON FOX BUSINESS
Representatives for Arizona Nutritional Supplements did not immediately respond to FOX Business' request for comment.
According to the FDA, the containers of protein powder, which were sold to Whole Foods Market locations nationwide, were mistakenly packaged with soy protein powder, creating an undeclared soy allergen.
As a result, the product may pose a serious or life-threatening risk to consumers who have an allergy or sensitivity to soy if consumed. However, no illnesses have been reported due to the recalled product.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
The recalled product, which was sold between March 24, 2021, and May 6, 2021, can be identified by the UPC 9948228764.
The recall notice comes after Whole Foods received a strict warning from federal health officials in December over its "pattern of receiving and offering for sale misbranded food products."
The FDA issued the warning letter issued to Whole Foods Market after discovering that undeclared allergens were the leading cause for dozens of its own recalls in the last year alone.
WHOLE FOODS ISSUES ALLERGY RECALL AFTER FDA'S STRICT WARNING TO IMPROVE FOOD LABELS
From October 2019 to November 2020, the company had recalled 32 food products "because the presence of major food allergens was not listed on the finished product labels," the FDA said. The agency cited similar recall patterns in previous years.
In its warning letter, the FDA said Whole Foods is "responsible" for investigating and determining the causes of its recent recalls in order to prevent their recurrence or other violations.
FDA ORDERS WHOLE FOODS TO IMPROVE ALLERGEN WARNINGS ON PRODUCTS
The company was also required to take "prompt action" to correct the violations if it didn't already, the FDA said in the letter. Failure to do so would potentially lead to further enforcement, including seizures or injunctions, the agency said.
However, three months after receiving the warning, Whole Foods already issued its own voluntary recall of its cranberry biscotti because of potentially undeclared pistachios.